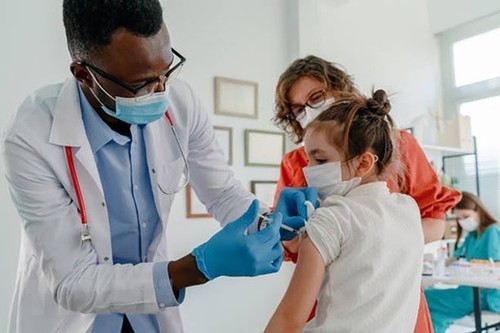
 A healthcare worker administers Pfizer's COVID-19 vaccine to children at a community health center in Los Angeles, California (USA). Photo: Getty Images/VNA A healthcare worker administers Pfizer's COVID-19 vaccine to children at a community health center in Los Angeles, California (USA). Photo: Getty Images/VNA
|
A day earlier, the US Food and Drug Administration expanded the emergency use authorizations for Moderna's vaccine to include children 6 months through 5 years and Pfizer/BioNTech's for children 6 months through 4 years.
The US CDC advisory panel says the shots help protect young children from possible hospitalization, death, and long-term complications that are still unknown due to the COVID-19 pandemic.